
Since this outbreak broke early last Saturday a week ago, I have learned far too much about Infant Botulism. I have now spoken with a half dozen experts and over a dozen parents of victims whose illnesses span from February 2025 to a few days ago. Interestingly, several of the early 2025 cases identified ByHeart formula as a product their child consumed before becoming sick. I know, correlation is not causation. But I intend to dig into them to find the facts.
We also know that the product was sold online by ByHeart, Amazon and perhaps other retailers outside the United States. Several countries from Canada to Trinidad and Tobago have posted warnings to their citizens.
However, taking a hard look at all the 2025 Infant Botulism with fresh eyes might well identify common denominators that may solve a larger problem and prevent the next one. I urge public health – local, state, federal and international – to take a second look at all Infant Botulism cases over the last year at least to determine any possible common sources.
The FDA and CDC, in collaboration with the California Department of Public Health (CDPH), Infant Botulism Treatment and Prevention Program (IBTPP), and other state and local partners, continue to investigate a multistate outbreak of infant botulism. Epidemiologic and laboratory data show that ByHeart Whole Nutrition infant formula might be contaminated with Clostridium botulinum, which is causing infant illness in multiple regions of the country.
Since the last update on November 11, 2025, nine new cases and one new state (Michigan) have been added to this investigation. All 9 cases were hospitalized and treated with BabyBIG®.
As of November 14, 2025, this outbreak includes 23 infants with suspected or confirmed infant botulism from 13 states – Arizona 3, California 3, Illinois 2, Kentucky 1, Michigan 1, Minnesota 2, North Carolina 2, New Jersey 1, Oregon 2, Pennsylvania 1, Rhode Island 1, Texas 3, Washington 2.
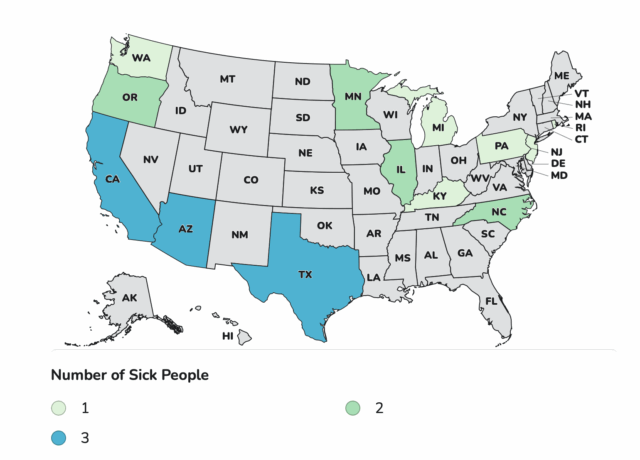
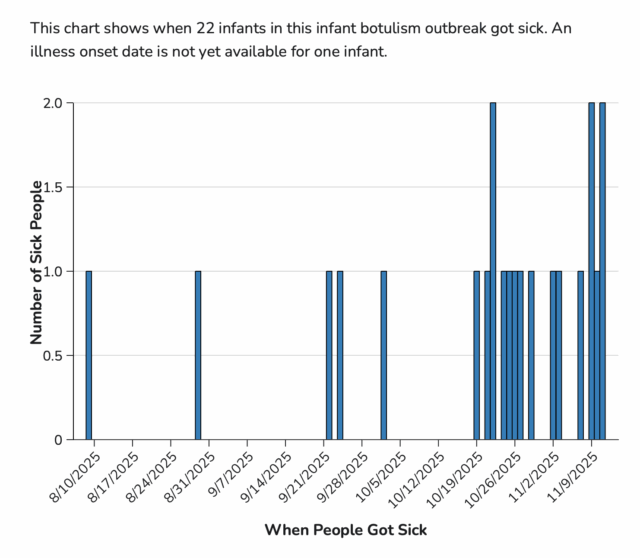
Laboratory confirmation for some cases is ongoing. For 22 cases with illness onset information available, illnesses started on dates ranging from August 9 to November 11, 2025 All 23 infants were hospitalized and treated with BabyBIG®. No deaths have been reported. For 22 infants with age and sex information available, they range in age from 16 to 200 days and 10 (45%) are female.
As part of this investigation, officials in several states have collected leftover infant formula for testing. On November 8, 2025, preliminary laboratory results reported by the California Department of Public Health suggest the presence of the bacteria that produce botulinum toxin in an open can of ByHeart infant formula (lot 206VABP/251131P2) that was fed to an infant with infant botulism. Additional testing is underway, and results are expected in the coming weeks. Detection of Clostridium botulinum in infant formula is difficult, and a negative test result does not rule out the presence of the bacteria in the product.